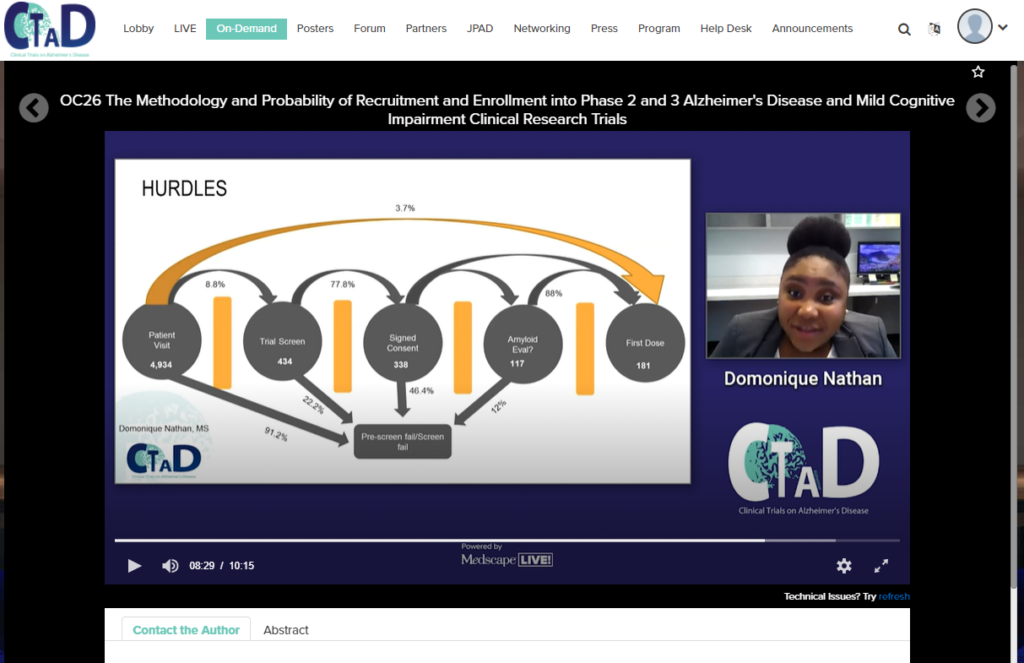
At the 13th Clinical Trials on Alzheimer’s Disease Conference, the staff of our Clinical Research Center presented “The Methodology and Probability of Recruitment and Enrollment into Phase 2 and 3 Alzheimer’s Disease and Mild Cognitive Impairment Clinical Research Trials”.
Excerpt from the abstract:
The rate of diagnosis for Alzheimer ’s Disease (AD) has continued to grow in the world’s geriatric population. Alzheimer’s Disease affects not only those diagnosed with the disease, but their family members and caregivers as well. Additionally, AD significantly impacts the economy, costing billions of dollars each year, estimated to increase to two trillion by 2030. An AD diagnosis is determined by a myriad of factors, some which include the presentation of symptoms, the presence of pathologies in fluid and imaging biomarkers. The progression of the disease is believed to be characterized by two pathologies: β-amyloid plaque and neurofibrillary tangles of hyperphosphorylated tau. Due to the limited understanding of the disease pathology, the potential for a cure is still widely debated. As a result, the majority of the clinical research trials investigating disease modifying therapies (DMTs), aim to slow the progression of the disease in order to increase the quality of life of those diagnosed. Among the ongoing clinical trials, a standard of 31,314 participants are needed to be recruited into all MCI and AD clinical trials across all sites. We performed a data analysis using a patient population that was seen for the last 10 years at Abington Neurological Associates’ (ANA) Clinical Research Center.